Covalent bonds are the strongest bonds that link atoms, with energies of bond formation on the order of -50 to -100 kcal/mole. The merging of electron orbitals that occurs during covalent bond formation places constraints on the angles at which atoms can be linked because orbital merging is energetically favorable only in certain directions. So, atoms sharing a covalent bond are held at fixed distances and within a range of favorable angles.
When two atoms share two electrons, the atoms are said to share a double bond. Double bonds are stronger than single bonds, with the result that double-bonded atoms are held closer together than those engaged in a single bond. Atoms in a double bond are not able to freely rotate about the bond axis as in a single bond, because sharing two electrons in their outer shells places additional constraints on the ways that their electron clouds can be oriented.
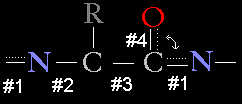
The peptide bond that links amino acids together in a protein chain is special in that it has the characteristics of a partial double bond. This is because the electrons shared in a double bond between the carboxy carbon and oxygen of one amino acid can resonate (delocalize) and can participate in the peptide bond linking the carboxy carbon to the amino nitrogen of the next amino acid.