I. IntroductionThe integrase family of DNA recombinases includes over sixty members identified by sequence similarity. Cre (shown at left) is a bacteriophage P1 member of the integrase family, catalyzing site-specific recombination between two, 34-base pair lox DNA sequences. In vivo, the function of Cre recombinase is to circularize the P1 genome during infection and maintain the genome in the monomeric state for cell division. Cre-lox recombination has also been extensively employed in in vivo genetic engineering applications in a variety of organisms. Before examining structural features of Cre Recombinase, it may be helpful to review the mechanism of Cre-induced, lox recombination as shown in Figure 1, below.
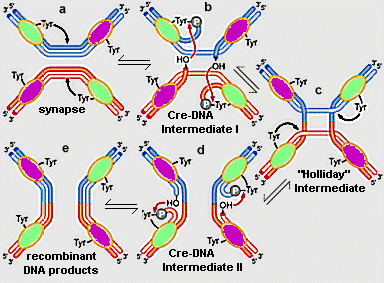
Figure 1. The site specific recombination reaction between two, lox DNA molecules (red and blue), catalyzed by Cre recombinase. (a) two Cre monomers bind to each lox site. A conserved, active site tyrosine (tyr324) from one of the monomers on each lox DNA molecule cleaves the DNA backbone, forming a covalent, 3' phosphotyrosine bond, leaving a free, 5' hydroxyl (OH) on one strand of each DNA double helix. (b) The 5' OH's perform a nucleophillic attack on the phophotyrosines from the partner DNA substrates, yielding a Holliday junction intermediate (c). (c)-(d) A second round of tyr324-catalyzed breakage, followed by strand joining reactions (nucleophillic attack of free OH's on phosphotyrosines) resolves the Holliday junction into recombinant products (e). Note that Cre monomers in the active conformation (green) and inactive conformation (purple) change sequentially in the process. Adapted from Figure 11-8, Watson, et al., 2003, Molecular Biology of the Gene. return to Cre-DNA Recombination Intermediates, below II. Cre Monomer StructureThe monomeric Cre protein shown at left contains amino terminal, linker, and carboxy terminal domains. The amino terminal domain comprises 5 alpha helices. Helices A and E are involved in Cre recombinase tetramer formation (see below). Helices B and D contact the lox DNA major groove. The carboxy terminal domain contains 9 alpha helices and 3 beta strands. Helix J, as well as several prominent loops, are involved in DNA interactions. Helix N protrudes from the domain and plays a role in intersubunit contacts (see below). The carboxy terminal domain harbors the Cre recombinase active site, consisting of arg173, his289, arg292, trp315, and the phosphate-attacking tyr324 (see Figure 1).
III. Cre-DNA Recombination IntermediatesLet's now explore the structure of intermediates in the Cre recombinase reaction. At left are shown two Cre monomers, each bound to a single lox half site of one DNA molecule. This corresponds to the top half of the schematic representation of Cre-DNA Intermediate I in Figure 1b. Note that the two monomers are in different conformations, only one of which is the active conformation. These conformations are switched as the reaction progresses (see below and Figure 1). Several of the helices mentioned in the previous section as being important in inter-Cre subunit interaction are positioned to promote association of the two monomers. Helix N from one subunit has one face buried in a hydrophobic pocket of the other monomer. Helices A and E from different monomers interact in a helix-helix interface. Other helices are positioned to interact with the Cre dimer bound to the partner lox DNA (not shown). Note that the B, D, and J helices from each monomer are inserted into the major grooves of the lox half sites. It can be seen that tyr324 from the active site of one monomer is covalently linked to the DNA through a 3' phosphate, a result of tyrosine-induced cleavage of the DNA backbone (see Figure 1b). The cleavage leaves a free 5' OH on one strand of DNA. The 5' OH will act as a nucleophile, attacking the phosphotyrosine from the partner DNA substrate (not shown), joining strands from opposite DNA helices. This yields a Holliday junction intermediate (see Figure 1b-c).
The structure of the Holliday junction intermediate in the Cre recombinase reaction is shown at left (compare to Figure 1c). The DNA of the the formerly separate lox sites is now joined through one recombinant strand. Cleavage and joining of the non-recombinant strands of the Holliday junction will produce fully recombinant DNA helices (Figure 1d-e). The Cre subunits are assembled into a tetramer on the Holliday junction DNA (Figure 1c), with each subunit bound to single lox half site. As noted above, the two subunits bound to each DNA molecule are in different conformations, with only one monomer on each DNA partner being active at one time. The pair of subunits that are active switches in the sequential one strand at a time mechanism of exchange (Figure 1).
IV. ReferencesGuo, F., Gopaul, D. N., van Duyne, G. D.: Structure of Cre recombinase complexed with DNA in a site-specific recombination synapse. Nature 389: 40-46 (1997). Gopaul, D. N., Guo, F., Van Duyne, G. D.: Structure of the Holliday junction intermediate in Cre-loxP site-specific recombination. EMBO J 17: 4175-4187 (1998). |
|